
The re-emitted light in the process is unlikely to be emitted in the same direction the absorbed photon originated from. This is because the atoms absorb light at a frequency dependent on the type of elements present in the sample. On the other hand, absorption can emit several colors coupled with blank lines. Colors in the prism are usually observed in these spectra. Here’s why: emission spectra can emit light that extends to the different ranges of the electromagnetic spectrum, thereby producing colored lines with low-energy radio waves to higher-energy gamma rays. In developing a picture, for instance, the emission spectrum is the colored photograph, while the absorption spectrum is the negative print. Quartz lamps are usually used in absorption, while burners are suitable for emission spectra.Īnother difference between the two spectra lies in the “print” output. Both need to have a light source, but these should vary in the two processes. In addition to this, absorption does not need the excitation of the ions or atoms, unlike emission spectra. In such cases, the spectrophotometer records the amount of wavelength emitted by the Earth in order for the scientist to determine the type of elements comprised by the solar system. When the giant flashlight is removed, the Earth then emits light in order for it to go back to its original state. 
The energy absorbed by the Earth is recorded in the absorption spectra. When a giant flashlight is directed towards the Earth (as an electron), the Earth becomes excited and moves up to the orbit of Neptune. The planets orbiting the Sun are the electrons. Think of it this way: the Sun is the center of the atom, consisting of photons and neutrons. In these processes, however, the scientist observes the amount of light or heat energy emitted by the photons of the atom that makes them go back to their original quantum. The spectrophotometer can record the amount of absorbed wavelength, and the scientist can then refer to the list of element characteristics to determine the composition of the sample gathered.Įmission spectra are performed with the same process of light subjection. When the light is directed towards atoms, ions, or molecules, the particles tend to absorb wavelengths that can excite them and cause them to move from one quantum to another. In the absorption spectra, he is supposed to observe how the electrons of the atoms absorb the electromagnetic energy from the light source. The scientist needs to have a list of values for both emission of absorption for each atom before subjecting the substance to spectroscopy.įor example, when the scientist discovers a sample from a far-flung area and aims to learn the composition of the matter, he may opt to subject the sample to emission or absorption spectroscopy. A spectrophotometer along with a light source is then needed in these processes. Both processes are geared to the observation of the electrons and photons when subjected to light. Absorption SpectraĪ chemist aiming to discover the elemental composition of a specific substance or solution can differentiate the atoms through emission and/or absorption spectroscopy. If we divide the observed value of A at λ max by the concentration of the sample ( c, in mol/L), we obtain the molar absorptivity, or extinction coefficient ( ε), which is a characteristic value for a given compound.Sodium Atomic Emission Spectrum Emission vs. The law is simply an application of the observation that, within certain ranges, the absorbance of a chromophore at a given wavelength varies in a linear fashion with its concentration: the higher the concentration of the molecule, the greater its absorbance. You most likely have performed a Beer – Lambert experiment in a previous chemistry lab. One of the most basic of these applications is the use of the Beer - Lambert Law to determine the concentration of a chromophore. UV-vis spectroscopy has many different applications in organic and biological chemistry. You can see that the absorbance value at 260 nm (A 260) is about 1.0 in this spectrum.Īpplications of UV spectroscopy in organic and biological chemistry To calculate absorbance at a given wavelength, the computer in the spectrophotometer simply takes the intensity of light at that wavelength before it passes through the sample (I 0), divides this value by the intensity of the same wavelength after it passes through the sample (I), then takes the log 10 of that number: 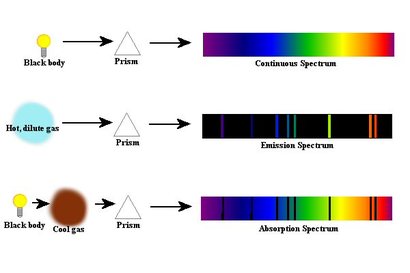
This contains the same information as the 'percent transmittance' number used in IR spectroscopy, just expressed in slightly different terms. Here we use a unitless number called absorbance, abbreviated 'A'.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
